Spectroscopy of microscopic areas on large samples
2030XL PRO™ Microspectrometer
The 2030XL PRO™ allows precise measurement of UV-visible-NIR range transmission, absorbance, reflectance, emission, and photoluminescent microspectra™ of features smaller than a micron. Remarkably, depending on the instrument configuration, it can handle samples of virtually unlimited size, making it exceptionally versatile for a wide range of applications.
Perfect Vision for Science™
Introduction
Features
Key Features*
UV-visible-NIR, Raman and PL microspectra™ of large samples.
|
UV-vis-NIR Microspectroscopy
Cutting edge microspectroscopy from the leaders
A fully integrated microspectroscopy unit that features a spectral range from the deep UV through the visible and into the near infrared. Simultaneous and direct imaging of both the sampling aperture and the sample makes for fast, accurate measurements. Featuring Scorpii™, SampleSafe™ and Lightblades™ technology, the 2030XL PRO™ gives you the ability to measure transmission, reflectance, polarization, Raman and fluorescence spectra of even sub-micron samples. CRAIC Technologies is also the only recognized source for NIST traceable microspectrometer standards. | 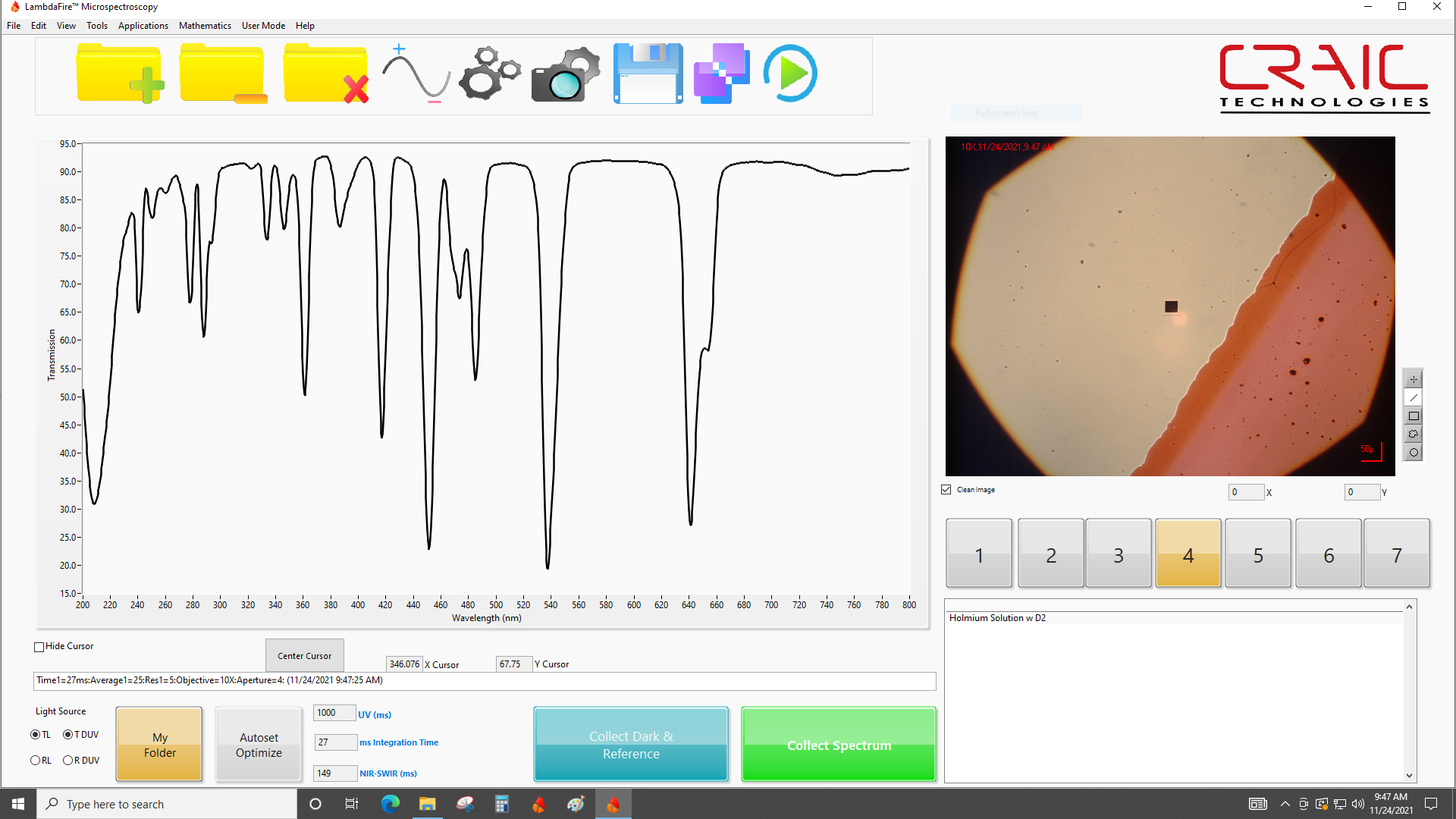 |
Raman Microspectroscopy
Flexible Raman Microspectroscopy
| When fitted with the CRAIC Apollo™ Raman spectrometer module, the 2030XL PRO™ is capable of Raman, resonance Raman and other types of measurements of microscopic samples. Modules include lasers, Raman spectrometers and the interface optics that allow you to collect high quality Raman spectra of your samples. | 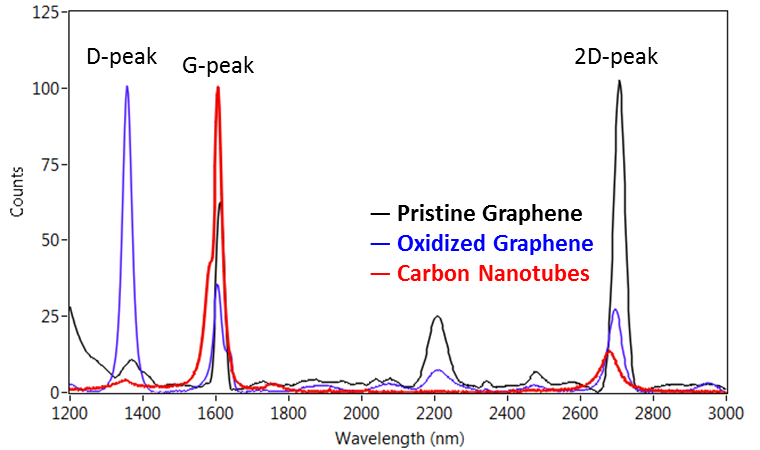 |
Photoluminescence
High sensitivity emission microspectroscopy & imaging
| The 2030XL PRO™ can be configured for fluorescence and photoluminescence spectra and images of microscopic samples. Featuring Lightblades™ technology and with excitation ranging from the deep UV to the near infrared and the ability to measure the emission in the same range, the 2030XL PRO™ is a powerful tool for microscale photoluminescence spectroscopy in the materials sciences, biology, geology and more. |  |
Polarization
Microspectroscopy & imaging of UV-visible-NIR polarization
| 2030XL PRO™ can be configured to acquire the polarization spectra and images of even the smallest samples. Featuring Lightblades™ technology and with a spectral range from the UV to the NIR, the 2030XL PRO™ polarization microspectroscopy capabilities are unparalleled. The spectra and images of birefringent and other types of samples with polarization characteristics can be acquired quickly and easily with this sophisticated system. | 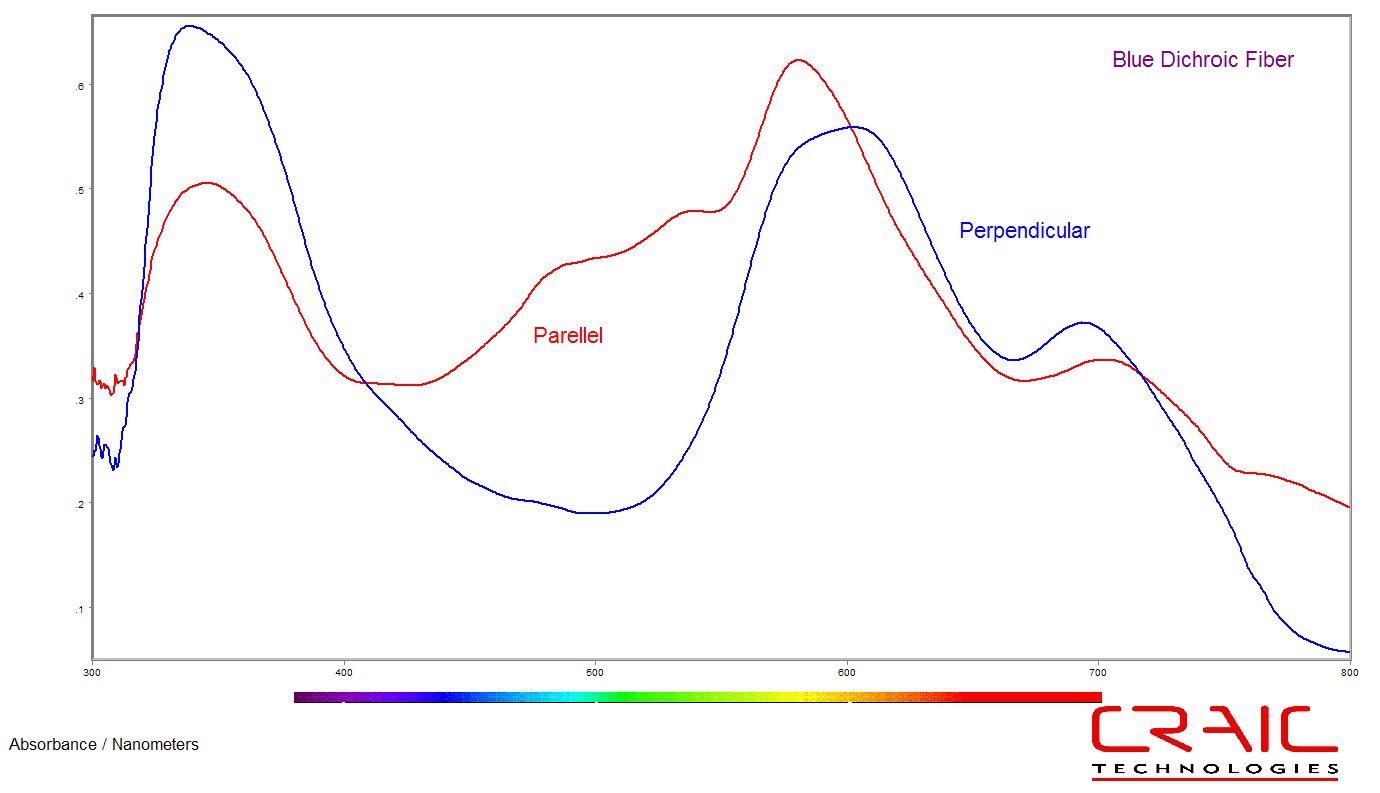 |
Spectral Surface Mapping
Combines hardware and software for automated spectral analysis and 5D mapping of samples with microscopic spatial resolution. 5D maps of the film thickness, transmission, reflectance, fluorescence, emission and Raman spectra of samples may be generated. | 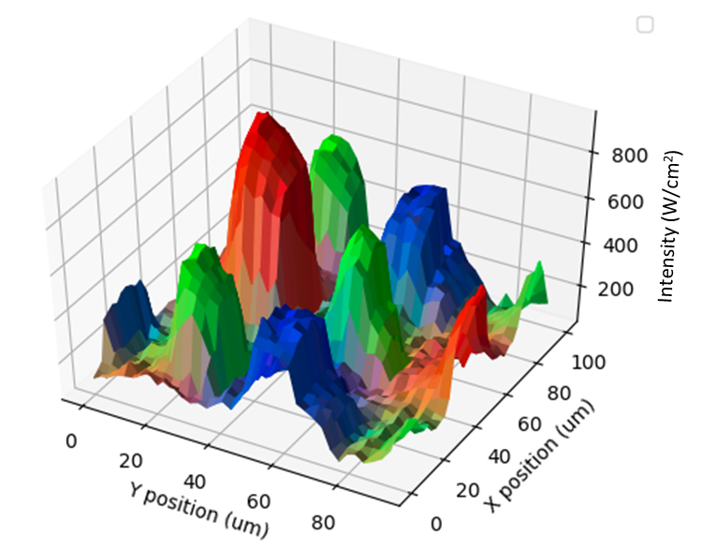 |
UV-vis-NIR Microimaging
Superior image quality from the UV to the NIR
| The 2030XL PRO™ includes a unique UV-visible-NIR microscope with research grade optics. Featuring high resolution digital imaging for color, UV and the NIR regions, the 2030XL PRO™ has sophisticated imaging software. This gives you the ability for both real time and image capture of samples by transmission, reflectance, polarization and fluorescence microscopy quickly and easily. |  |
Applications
Applications
- Semiconductor Film Thickness
- MEMS devices
- Photovoltaic Panels
- Colorimetry of flat panel displays
- Intensity mapping of flat panel displays
- Microspectra™ of Questioned Documents
- Microspectra™ of Art Works
- or any other large scale samples...
Support
Support
CRAIC Technologies provides service and support for it's instruments worldwide. CRAIC Technologies service engineers offer instrument repair, maintenance, training and technical support for all aspects of CRAIC Technologies products.

The 2030XL PRO™ Microspectrometer can take spectra and images of microscopic samples from the deep ultraviolet to near infrared with one seamless operation. It can acquire microspectra™ and images in absorbance, reflectance, and fluorescence. The microspectrometer is offered with both the DirecVu™ to view samples by eye as well as with a high resolution UV-visible-NIR digital imaging system.
The lit microscope base and the lit octagonal optical head are trademarks of CRAIC Technologies, Inc. CRAIC Technologies, 2030PV™, FLEX™, 508PV™, Apollo™, GeoImage™, rIQ™, Lambdafire™, Lightblades™ and "Perfect Vision for Science" are all trademarks of CRAIC Technologies, Inc. Instrument features offered depend upon instrument configuration. Features listed here may not be present in some configurations.
*Features and specifications depend upon instrument configuration. Specifications subject to change without notice.
UV-visible-NIR microscopes, UV-visible-NIR microspectrometers and Raman microspectrometers are general purpose laboratory instruments. They have not been cleared or approved by the European IVD Directive, the United States Food and Drug Administration or any other agency for diagnostic, clinical or other medical use.

